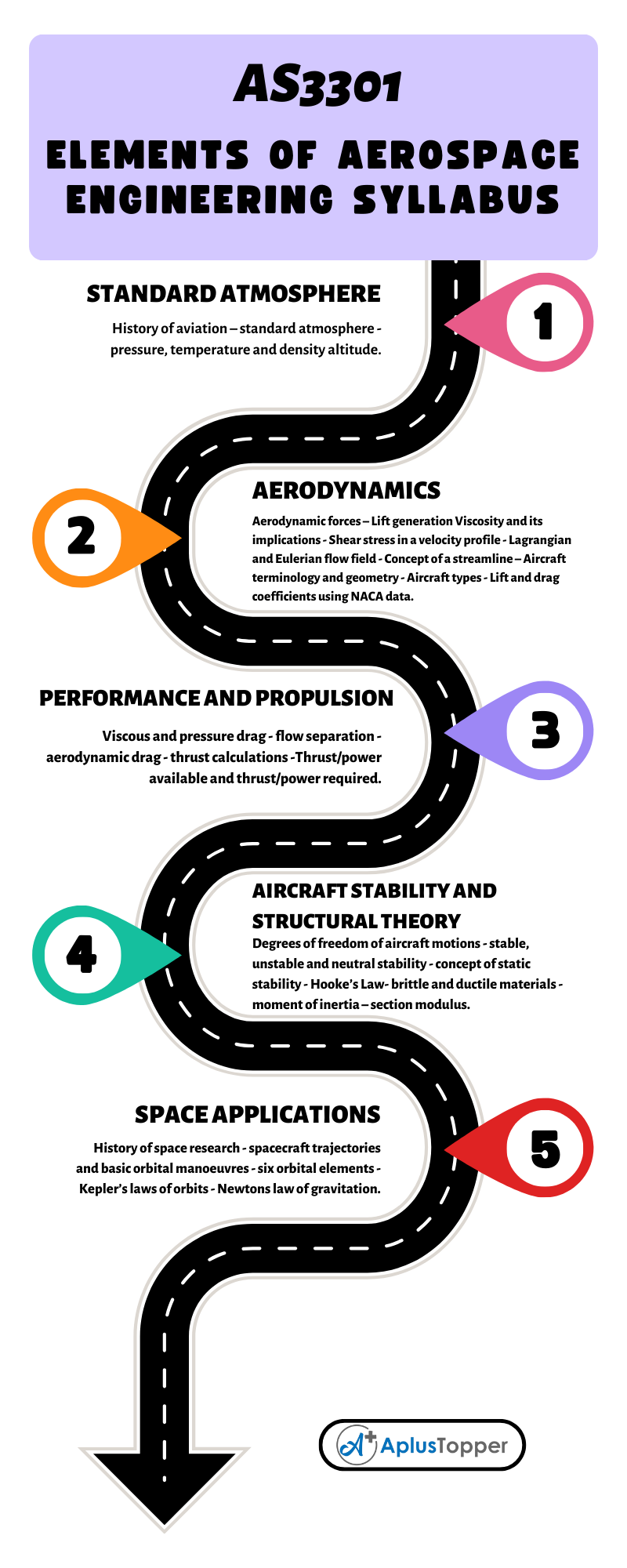
Redox Reactions by Transfer of Electrons at a Distance
Redox Reactions by Transfer of Electrons at a DistanceIn all redox reactions, electrons are transferred from the reducing agent to the oxidising agent.When the reducing and oxidising agents are mixed together as in the previous reactions, the transfer of…
Redox reaction in the displacement of metals from its salt solution
Redox reaction in the displacement of metals from its salt solutionGenerally, metals are good electron donors and therefore are good reducing agents. However, different metals have different strength as reducing agents.The strength of metals as reducing …
Changing of iron(II) ions to iron(III) ions and vice versa
Changing of iron(II) ions to iron(III) ions and vice versaIron exhibits two oxidation numbers (a) +2 as iron(II) ion, Fe2+ (b) +3 as iron(III) ion, Fe3+An aqueous solution containing iron(II) ions, Fe2+ is pale green in colour, whereas that containing ir…