Plus Two Chemistry Chapter Wise Questions and Answers Chapter 13 Amines is part of Plus Two Chemistry Chapter Wise Questions and Answers. Here we have given Plus Two Chemistry Chapter Wise Questions and Answers Chapter 13 Amines.
| Board | SCERT, Kerala |
| Text Book | NCERT Based |
| Class | Plus Two |
| Subject | Chemistry Chapter Wise Questions |
| Chapter | Chapter 13 |
| Chapter Name | Amines |
| Category | Kerala Plus Two |
Kerala Plus Two Chemistry Chapter Wise Questions and Answers Chapter 13 Amines
Plus Two Chemistry Amines One Mark Questions and Answers
Question 1.
The reaction of aniline with benzoyl chloride gives
(a) benzoin
(b) benzanilide
(c) benzalaniline
(d) benzamide
Answer:
(b) benzanilide
Question 2.
Nitromethane on reaction with H2/Pd gives ____________
Answer:
Ethanamine (CH3CH2NH2)
Question 3.
Say TRUE or FALSE
Only aliphatic primary amines give a foul smelling compound with CHCl3 and alcoholic potash.
Answer:
False
Question 4.
When benzene diazonium salt solution is treated with KI ___________ is formed.
Answer:
lodo benzene (C6H5I)
Question 5.
Identify ‘Z’ in the sequence:
![]()
(a) Nitrobenzene
(b) Benzene
(c) Fluorobenzene
(d) Phenol
Answer:
(a) Nitrobenzene
Question 6.
Amine that cannot be prepared by Gabriel Phthalimide Synthesis is _________
Answer:
Aniline
Question 7.
Phenyl isocyanide, is prepared from aniline by ________
Answer:
Carbylamine reaction
Question 8.
Secondary amines can be prepared by ___________
Answer:
Reduction of nitro compounds
Question 9.
By which process aniline can be purified ___________
Answer:
Steam distillation
Question 10.
A primary amine that can be obtained both by the reduction of cyanides and amides is ___________
(a) methyl amine
(b) benzylamine
(c) aniline
(d) isopropylamine
Answer:
(b) benzylamine
Question 11.
The amine which will not liberate nitrogen on reaction with nitrous acid is __________
Answer:
t-butyl amine
Plus Two Chemistry Amines Two Mark Questions and Answers
Question 1.
Ammonia is less basic than aniline.
- What is the reason for low basic character of aniline compared to ammonia?
- Draw the resonating structures of aniline.
Answer:
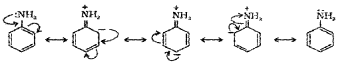
1. In aromatic amines the lone pair on nitrogen is in conjugation with benzene ring due to resonance effect and thus making it less available for protonation. Hence basic character is less than that of ammonia.
2.
Question 2.
Two compounds are functional isomers of each other. On reduction one gives primary amine and the other gives secondary amine.
- Identify the class of compounds.
- Explain the reduction reaction.
Answer:
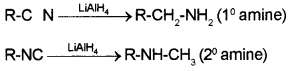
- Cyanides and Isocyanides.
- When cyanides are reduced using LiAlH4 primary amines are obtained. Whereas when isocyanides are reduced using LiAlH4 secondary amines are obtained.
Question 3.
When alkyl halide is treated with alkali metal cyanide, cyanides are obtained as the major product. Assume that AgCN is used instead of NaCN.
- What will be the product?
- Justify.
Answer:
- Isocyanide
- Silver cyanide is predominantly covalent. Hence nitrogen atom is free to donate electron pair forming isocyanide as the main product.
Question 4.
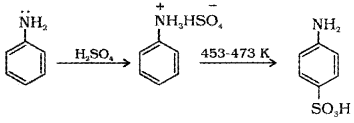
How is sulphanilic acid prepared from aniline?
Answer:
Aniline reacts with concentrated sulphuric acid to form anilinium hydrogensulphate which on heating with sulphuric acid at 453-473 K produces sulphanilic acid.
Question 5.
Write chemical equations for the preparation of benzene, fluorobenzene, and nitrobenzene from benzenediazonium chloride.
Answer:
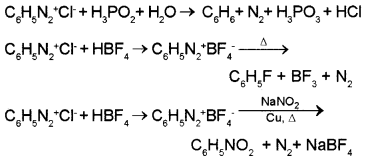
Question 6.
- What are Schiff s bases?
- How are they formed?
Answer:
1. Schiff’s bases are substituted imines.
2. These are formed when 1° amines are treated with carbonyl compounds.
![]()
Question 7.
What are the products formed by the reaction of ethanolic NH3 with C2H5CI? Write the chemical equation.
Answer:
A mixture of ethanamine, N-ethylethanamine and N,N-diethylethanamine are formed
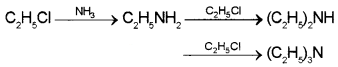
Question 8.
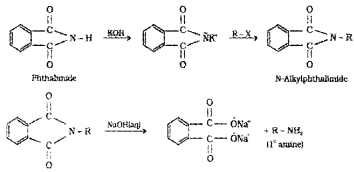
Gabriel phthalimide synthesis is preferred for synthesising primary amines.
Answer:
Gabriel phthalimide synthesis involves the conversion of alkyl halide (R – X) to 1° amine (R – NH2). Ammonolysis of R – X, on the other hand give 2°, 3° and quaternary salt as other byproducts. Hence, for the production of pure 1° amines, Gabriel phthalimide synthesis is preferred to ammonolysis reaction.
Plus Two Chemistry Amines Three Mark Questions and Answers
Question 1.
Nitrogen containing functional groups are classified into different types.
- Which are they?
- Explain one method to prepare cyanide.
- Give chemical equations.
Answer:
- Nitro compounds, Amines, Cyanides, Isocyanides and Diazo compounds.
- When alkyl halides on heating with alcoholic KCN or NaCN cyanide is obtained.
- R – X + KCN → R-CN + KX. e.g. CH3 – CH2 -Br + KCN → CH3CH2CN + KBr
Question 2.
Consider the following chemical equations:
- a) Identify ‘A’ and ‘B’.
- b) Name the reactions.
- c) Give any one method to prepare beznene diazonium chloride.
Answer:
1. A – LiAIH4 B – Br2/KOH
2.
- i) is reduction of acid amide.
- ii) is Hoffmann’s bromamide degradation reaction.
3. Benzene diazonium chloride can be prepared by diazotization of aniline. It can be done by treating aniline with sodium nitrite and HCI at 273-278 K.
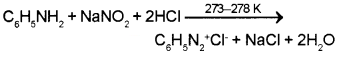
Question 3.
Chlorobenzene can be prepared from benzene diazonium chloride in two ways.
- a) Do you agree with it?
- b) Which are the two ways?
- c) Write the chemical equation for the reactions.
Answer:
1. Yes.
2. By Sandmeyer’s reaction and Gattermann’s reaction.
3. Sandmeyer’s reaction
![]()
Gattermann’s reaction
![]()
Question 4.
Match the following:
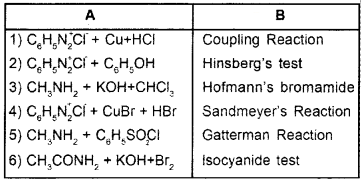
Answer:
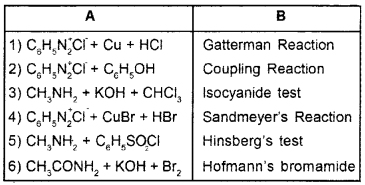
Question 5.
Diazonium salts are very important class of compounds used for synthesis of variety of aromatic compounds.
- How is nitrobenzene prepared from diazonium salt?
- Give an example for coupling reaction.
- Give an important use of diazonium salt.
Answer:
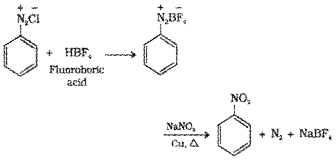
1. Benzene diazonium chloride is treated with fluoroboric acid to get benzenediazonium fluoroborate which when heated with aqueous sodium nitrite solution in the presence of copper, nitrobenzene is formed.
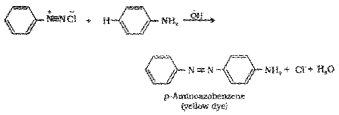
2. When benzenediazonium chloride is coupled with aniline in acid medium, p-aminoazobenzene (an yellow azo dye) is formed.
3. It can be used as intermediate for the synthesis for many organic compounds.
Question 6.
An aromatic compound ‘A’ on treatment with aqueous ammonia and heating forms compound ‘B’ which on heating with Br2 and KOH forms a compound ‘C’of molecular formula C6H7N. Write the structures and IUPAC names of compounds A, B, and C.
Answer:
Hoffman bromide reaction
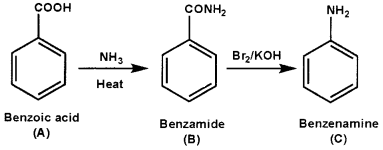
Question 7.
Write the IUPAC names of the following compounds and classify them into primary, secondary and tertiary amines.
- C6H5NHCH3
- (CH3CH2)2NCH3
- m-BrC6H4NH2
Answer:
- N-Methylbenzenamine (2° amine)
- N-Ethyl-N-methylethanamine (3° amine)
- 3-Bromobenzenamine (1° amine)
Plus Two Chemistry Amines Four Mark Questions and Answers
Question 1.
C6H5NH2 gives a foul smelling compound ‘A’ with chloroform in presence of KOH (alcoholic).
- What is compound ‘A’?
- Write the chemical equation for the reaction.
Answer:
a) ‘A’ is phenyl isocyanide (or phenyl carbylamine)
b) Carbyl amine reaction
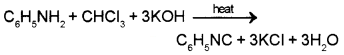
Question 2.
1. In a group discussion, a student argued that alkyl amines are more basic than NH3.
- Is it correct?
- Justify your answer with suitable explanation.
2. Arrange the following amines in the decreasing order of basic strength in aqueous solution.
CH3NH2, (CH3)2NH, NH3, (CH3)3N Justify.
Answer:
1.
- Yes.
- Electron realeasing inductive effect (+l) of alkyl groups increases the availability of lone pair on nitrogen.
2. (CH3)2NH > CH3NH2 > (CH3)3N > NH3
Inductive effect is maximum for 3° amines. At the same time steric hindrance is maximum for 3° amines. An interplay of the inductive effect, solvation effect and steric hindrance of the alkyl group decides the basic strength of alkylamines.
Question 3.
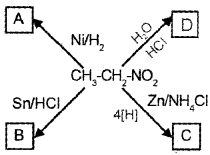
a) Watch the above diagram and fill the labelled boxes A, B, C, and D.
b) If you are treating B with nitrous acid, predict the product that can be formed.
Answer:
a)
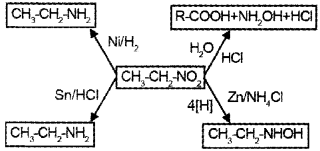
b) CH3 CH2 – OH
Question 4.
a) Benzenediazonium chloride is a very important compound in organic chemistry. Give the structure. How it is prepared?
b) How phenol and iodobenzene are prepared from the above compound?
Answer:
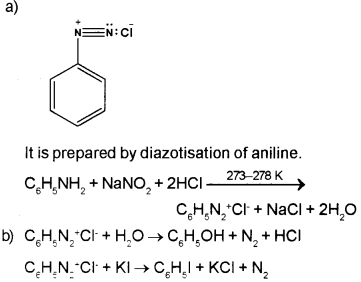
Question 5.
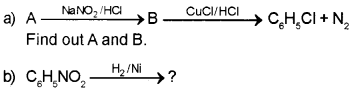
Answer:
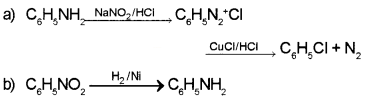
Question 6.
a) Arrange the following in the increasing order of basic strength.
C6H5NH2, C6H5N(CH3)2, (C2H5)2NH, CH3NH2
b) How will you convert aniline to phenol?
Answer:
a) C6H5NH2 < C6H5N(CH3)2 < CH3NH2 < (C2H5)2NH
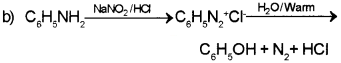
Question 7.
- How is nitrous acid prepared in the laboratory?
- A student treated methylamine and aniline separately with nitrous acid. What are the products formed in each case? Give chemical equations.
Answer:
1. By treating hydrochloric acid with sodium nitrite,
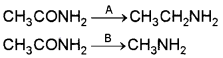
2. Methyl amine reacts with nitrous acid to form methyldiazonium salt which being unstable, liberates nitrogen gas quantitatively and forms methanol.
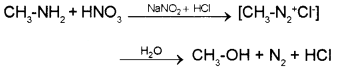
Aniline reacts with nitrous acid at low temperature (273-278 K) to form benzenediazonium salt, a very important compound used in synthetic organic chemistry.
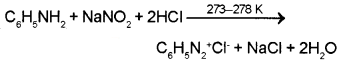
Question 8.
Give one chemical test to distinguish between the following pairs of compounds.
- Methylamine and dimethylamine
- Secondary and tertiary amines
- Ethylamine and aniline
- Aniline and benzylamine
- Aniline and N-methylaniline
Answer:
1. Methyl mine is a 1° amine and so it gives the carbylamine test. When methylamine is warmed with chloroform and alcoholic solution of KOH, foul smelling methyl isocyanide is formed.
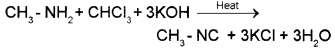
Dimethylamine is a 2° amine and hence it does not answer carbylamine test. Or they can be distinguished by Hinsberg’stest.
2. Secondary and tertiary amines can be distinguished by Hinsberg’s test.
Secondary amines react with Hinsberg’s reagent (benzene sulphonyl chloride) to form N, N- dialkyl benzene sulphonamide which is insoluble in alkali.
Tertiary amines do not react with Hinsberg’s reagent.
3. Ethylamine is an aliphatic 1° amine. When treated with HNO2 (NaNO2 and HCI) it forms ethanol with liberation of N2 gas.
CH3 – CH2 – NH2 + HNO2 → CH3 – CH2 – OH + N2 + H2O
Aniline is an aromatic 1° amine. When treated with HNO2 at low temperature (273 – 278 K) it gives benzene diazonium chloride which undergoes coupling reaction with phenol to form an orange azo dye.
C6H5NH2 + HNO3 + HCI → C6H5N2+ + Cl– + 2H2O
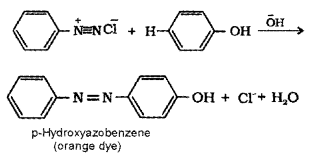
4. Aniline forms azodye with benzene diazonium chloride but benzyl amine does not.
5. Aniline being a primary amine gives carbylamine test while N-methylaniline being a 2° amine does not answer carbylamine test.
Question 9.
Write short notes on the following:
- Carbylamine reaction.
- Hoffman’s bromamide reaction.
Answer:
1. Methyl amine is a 1° amine and so it gives the carbylamine test. When methylamine is warmed with chloroform and alcoholic solution of KOH, foul smelling methyl isocyanide is formed.
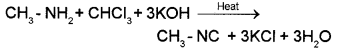
Dimethyl amine is a 2° amine and hence it does not answer carbylamine test. Or they can be distinguished by Hinsberg’stest.
2. When an acid amide is heated with Br2 and an aqueous or ethanolic solution of alkali, a primary amine containing one carbon less than the initial amide is obtained. This is called Hoffmann’s bromamide degradation reaction.
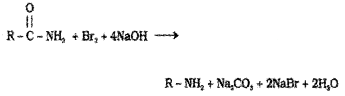
Question 10.
Write the IUPAC names of the following compounds and classify them into primary, secondary and tertiary amines.
- (CH3)2CHNH2
- CH3(CH2)2NH2
- CH3NHCH(CH3)2
- (CH3)3CNH2
Answer:
- Propan-2-amine (1° amine)
- Propan-1-amine (1° amine)
- N-Methylpropan-2-amine (2° amine)
- 2-Methylpropan-2-amine (1° amine)
Question 11.
Account for the following:
- pKb of aniline is more than that of methylamine.
- Ethylamine is soluble in water whereas aniline is not.
Answer:
1. In CH3– NH2, the +l effect of methyl group increases the electron density around N atom and it increases the electron releasing tendency of the molecule. In C6H5 – NH2 the resonance effect causes delocalisation of lone pair over the ring and thereby decreases its basic strength. Since aniline is less basic than methylamine, its pKb value is greater than that of CH3NH2.
2. Solubility of ethylamine in water is attributed to its ability to form hydrogen bonds with water molecules. In aniline the non-polar hydrocarbon part (the ring skeleton) is relatively larger and therefore, has no interactions with polar water molecules. It also decreases the tendency of hydrogen bonding with water molecules. Hence, aniline is not soluble in water.
Question 12.
Account for the following:
- Methylamine in water reacts with ferric chloride to precipitate hydrated ferric oxide.
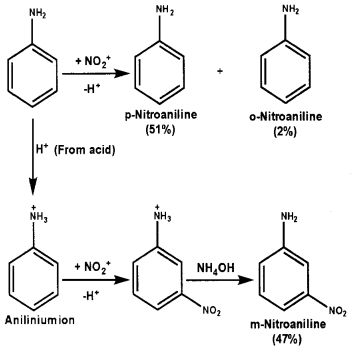
- Although amino group is o- and p- directing in anomatic electrophilic substitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline.
Answer:
1. Methyl amine, being more basic than water exists in water as N-methylammonium hydroxide which reacts with ferric chloride to form hydrated ferric oxide.
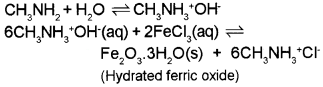
2. Aniline being a base mostly gets protonated in the presence of acids to form anilinium ions (NH3+). For electrophlic ring substitution, – NH2 group is activating and ortho and para directing whereas – NH3+ is deactivating and meta directing. In aniline, the probability of NO2+ attack at para position is relatively more because of steric hinderance at ortho position. Anilinium ion, the attack of NO3+ mostly occurs at meta position.
Question 13.
Account for the following:
- Aniline does not undergo Friedel-Crafts reaction.
- Diazonium salts of aromatic amines are more stable than those of aliphatic amines.
Answer:
1. Aniline is a base due to electron donating nature of lone pair on N atom of -NH2 goup. Aniline therefore, forms salt with AICI3 which is Lweis acid and a catalyst used in Friedel Craft’s reaction.
![]()
So the catalyst will not be available to produce electrophile.
2. Arenediazonium ion is resonance stabilised whereas no resonance stabilisation occurs in alkyl diazonium ion.
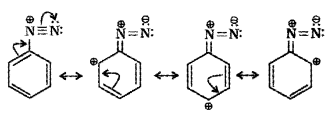
Plus Two Chemistry Amines NCERT Questions and Answers
Question 1.
Write the IUPAC names of the following compounds and classify them into primary, secondary and tertiary amines.
- (CH3)2CHNH2
- CH3(CH2)2NH2
- CH3NHCH(CH3)2
- (CH3)3CNH2
- C6H5NHCH3
- (CH3CH2)2NCH3
- m-BrC6H4NH2
Answer:
- Propan-2-amine (1° amine)
- Propan-1-amine (1° amine)
- N-Methylpropan-2-amine (2° amine)
- 2-Methylpropan-2-amine (1° amine)
- N-Methylbenzenamine (2° amine)
- N-Ethyl-N-methylethanamine (3° amine)
- 3-Bromobenzenamine (1° amine)
Question 2.
Give one chemical test to distinguish between the following pairs of compounds.
- Methylamine and dimethylamine
- Secondary and tertiary amines
- Ethylamine and aniline
- Aniline and benzylamine
- Aniline and N-methylaniline
Answer:
1. Methyl amine is a 1° amine and so it gives the carbylamine test. When methylamine is warmed with chloroform and alcoholic solution of KOH, foul smelling methyl isocyanide is formed.
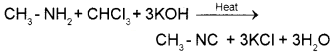
Dimethyl amine is a 2° amine and hence it does not answer carbylamine test. Or they can be distinguished by Hinsberg’stest.
2. Secondary and tertiary amines can be distinguished by Hinsberg’stest.
Secondary amines react with Hinsberg’s reagent (benzene sulphonyl chloride) to form N, N- dialkyl benzene sulphonamide which is insoluble in alkali.
Tertiary amines do not react with Hinsberg’s reagent.
3. Ethyl amine is an aliphatic 1° amine. When treated with HNO2 (NaNO2 and HCI) it forms ethanol with liberation of N3 gas.
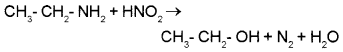
Aniline is an aromatic 1° amine. When treated with HNO2 at low temperature (273 – 278 K) it gives benzene diazonium chloride which undergoes coupling reaction with phenol to form an orange azo dye.
C6H5NH2 + HNO3 + HCI → C6H5N2++Cl– + 2H2O
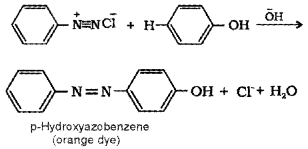
4. Aniline forms azodye with benzene diazonium chloride but benzyl amine does not.
5. Aniline being a primary amine gives carbylamine test while N-methylaniline being a 2° amine does not answer carbylamine test.
Question 3.
How will you convert:
(i) Ethanoic acid into methanamine
(ii) Hexanenitrile into 1-aminopentane
Answer:
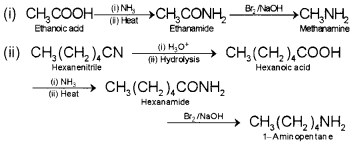
Question 4.
How will you convert:
(i) Methanol to ethanoic acid
(ii) Ethanamine into
Answer:
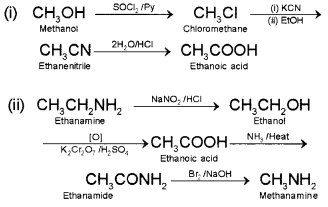
Question 5.
How will you convert:
(i) Ethanoic acid into propanoic acid
(ii) Nitromethane into dimethylamine
Answer:
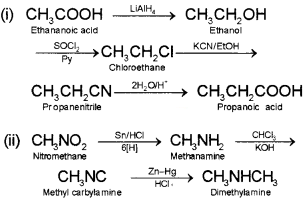
Question 6.
An aromatic compound ‘A’ on treatment with aqueous ammonia and heating forms compound ‘B’ which on heating with Br2 and KOH forms a compound ‘C’ of molecular formula C6H7N. Write the structures and IUPAC names of compounds A, B, and C.
Answer:
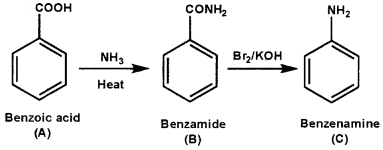
Question 7.
Write short notes on the following:
- Carbylamine reaction
- Diazotisation
- Hoffmann’s bromamide reaction
- Coupling reaction
- Gabriel phthalimide synthesis.
Answer:
1. Carbylamine reaction
Primary amines (both aliphatic and aromatic) when heated with chloroform and ethanolic potassium hydroxide form isocyanides (also known as carbylamines) which have a foul smell. This reaction is called carbylamine reaction and is used as a test for primary amines. Secondary and tertiary amines do not show this reaction.
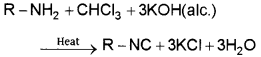
2. Diazotisation:
Aromatic amines react with nitrous acid (HNO2) at low temperature (273-278 K) to form diazonium salts. The process is known as diazotisation eg.
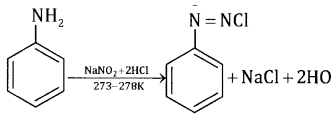
3. Hofmann’s bromamide reaction:
In this reaction, an acid amide is heated with Br2 and aq. NaOH when 1° amine having one carbon atom less is produced. This involves migration of alkyl or aryl group from carboxyl carbon of the amide to nitrogen atom of the amine. The amine so formed contains one carbon less than that present in amide.
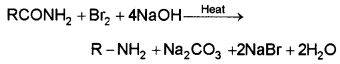
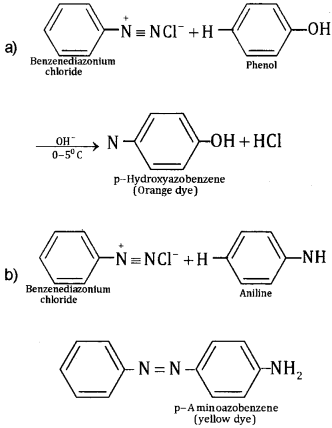
4. Coupling reaction:
Diazonium salts react with phenol and amines to give azo compounds which have an extended conjugate system having both the aromatic rings joined by -N = N – bond. eg.
5. Gabriel phthalimide synthesis:
This method is used for preparing only 1° amines. In this method, phthalimide is treated with alcoholic KOH to give potassium phthalimide, which is treated with alkyl halide or benzyl halide to form N- alkyl or aryl phthalimide. The hydrolysis of N- alkyl phthalimide with 20% HCI under pres¬sure or refluxing with NaOH gives 1° amines.
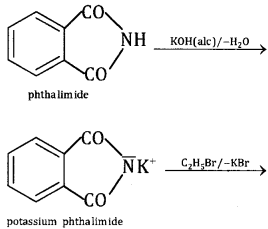
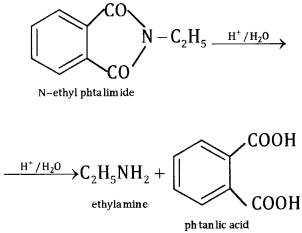
The more convenient method is by the treatment of alkyl phthalimide with hydrazene.
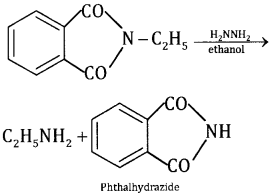
Phthalic acid can again be converted into phthalimide and is used again and again. This method is very useful because it gives pure amines.
Question 8.
Give plausible explanation for each of the following:
- Why are amines less acidic than alcohols of comparable molecular masses?
- Why are primary amines higher boiling than tertiary amines?
- Why are aliphatic amines stronger bases than aromatic amines?
Answer:
1. Loss of proton from amines give amide ion whereas loss of a proton from alcohol gives an alkoxide ion.
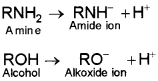
Since O is more electronegative than N, therefore, RO– can accomodate the -ve charge more eaisly than RNH–. Consequently, RO– is more stable than RNH–. Thus, alcohols are more acidic than amines.
2. Primary amines (RNH2) have two hydrogen atoms on the N atoms and therefore, form intermolecular hydrogen bonding
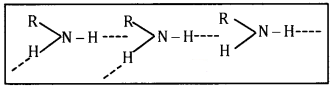
Tertiary amines (R3N) do not have hydrogen atoms on the N atom and therefore, these do not form hydrogen bonding in primary amines, they have higher boiling points than tertiary amines of comparable molecular mass. For example, boiling point of n-butylamine is 351 K while that of tert-butylamine is 319K.
3. Both arylamines and alkalamines are basic in nature due to the presence of lone pair on N-atom. But arylamines are less basic than alkyamines. For example, aniline is less basic than ethylamine as shown by Kb values:
Ethylamine: Kb = 4.7 × 10-4
Aniline: Kb =4.2 × 10-10
The less basic character of aniline can be explained on the basis of aromatic ring present in aniline. Aniline can have the following resonating structures:
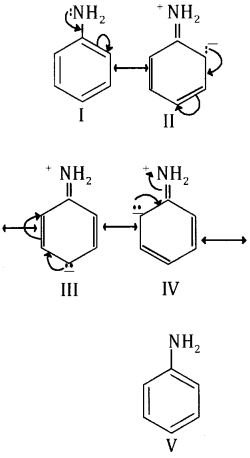
It is clear from the above resonating structures that three of these (II, III and IV) acquire some positive charge on N atom. As a result, the pair of electrons become less available for protonation. Hence, aniline is less basic than ethyl amine in which there is no such resonance.
We hope the Kerala Plus Two Chemistry Chapter Wise Questions and Answers Chapter 13 Amines help you. If you have any query regarding Kerala Plus Two Chemistry Chapter Wise Questions and Answers Chapter 13 Amines, drop a comment below and we will get back to you at the earliest.
