Plus Two Chemistry Chapter Wise Previous Questions Chapter 13 Amines is part of Kerala Plus Two Chemistry Chapter Wise Previous Year Questions and Answers. Here we have given Plus Two Chemistry Chapter Wise Questions and Answers Chapter 13 Amines.
Kerala Plus Two Chemistry Chapter Wise Previous Questions Chapter 13 Amines
Question 1.
Aromatic amines are important synthetic intermediates. (March – 2010)
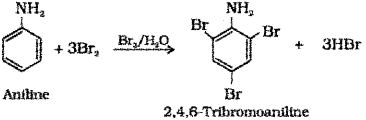
i) What are the products obtained when aniline is treated with bromine water?
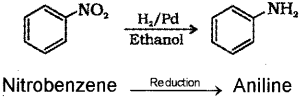
ii) How will you convert nitrobenzene to aniline?
iii) Write down the Isocyanide test for the primary amines.
Answer:
i) Aniline reacts with bromine water at room temperature to give a white precipitate of 2,4,6 tribromoaniline.
ii) Nitrocompounds on reduction gives amines.
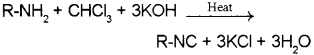
iii) Primary amines on heating with chloroform and ethanolic KOH, foul-smelling substances known as isocyanides or carbylamines are formed.
Question 1.
Benzene sulphonyl chloride and aqueous NaOH can be used to distinguish three classes of amines such as primary, secondary and tertiary. (Say – 2010)
a) Name the above test.
b) How will you distinguish the above amines using this test?
c) Give the reactions and justifications,
Answer:
a) Hinsberg test. C6H5SO2Cl
b) 10 Amine + Hinsberg reagent → a compound soluble in alkali
20 Amine + Hinsberg reagent → A compound which is insoluble in alkali
30 Amine +Hinsberg reagent → no reaction.
c) The hydrogen attached to nitrogen in sulphonamide is strongly acidic due to the presence of strong electron withdrawing sulphonyl group. Hence, it is soluble in alkali.
Secondary amine react with benzene salphonyl chloride to form N, N-dialkylbenzene- sulphon-amide, which is insoluble in alkali. This is be?cause it does not contain any hydrogen attached to nitrogen atom and is not acidic.
Tertiary amines do not react with benzene salphonyl chloride, because they do not possess any replacable hydrogen.
Question 1.
Amines are versatile functional group useful in the preparation of many organic compounds. How can you convert? (March – 2011)
OR
a) A student tried to prepare p-nitroaniline by nitrating Aniline with Conc. HNO3 – Coric. H2SO4 mixture, but he got only m-nitro aniline. Why?
b) Explain how he should proceed to get pnitroaniline from Aniline.
Answer:
a) In the strongly acidic medium aniline is protonated to form anilium ion which is meta directing.
b) The -NH2 group in aniline should be protected by acetylation by treating it with acetic anhydride. The acetanilide formed ¡s subjected to nitration to get p-Nitroacetanilide which on hydrolysis gives p-Nitroaniline.
Question 1.
Primary, secondary and teritary amines can be distinguished using Hinsberg’s reagent. (March – 2012)
i) What is Hinsberg’s reagent?
ii) How will you distinguish primary, secondary and tertiary amines using Hinsberg’s reagent?
Answer:
i) Benzene salphonyl chloride (C6H5SO2Cl)
ii) a) The reaction of C6H5SO2Cl with primary amine yields N-alkyl benzenesulphonamide which is soluble in alkali
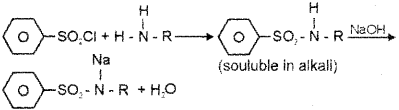
The hydrogen attached to nitrogen in suiphonamide is strongly acidic due to the presence of strong electron withdrawing suiphonyl group. Hence, it is soluble in alkali.
b) Secondary amine read with benzene salphonyl chloride to form N, N-dialkylbenzene sulphonamide, which is insoluble in alkali. This is because it does not contain any hydrogen attached to nitrogen atom arid is not acidic.
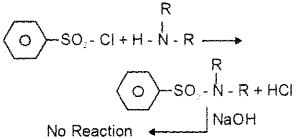
c) Tertiary amines do not react with benzene salphonyl chloride, because they do not pos-sess any replacable hydrogen.
Question 1.
a) Carbyl amines have an offensive smell. (Say – 2012)
i) Write the carbyl amine reaction.
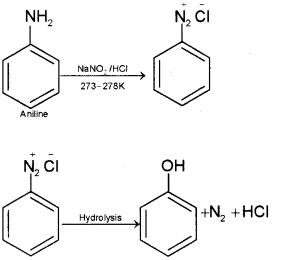
ii) How will you convert aniline into phenol?
b) How will you convert an amide into the following? 0 An amine with one carbon atom less than that
of the amide.
ii) An amine containing the same number of car bon atoms as that in the amide.
Answer:
a) i) Aliphatic and aromatic primary amines on heating with chloroform and ethanolic potas skim hydroxide form foul smelling substances called isocyanides or carbylamines. This reaction is known as carbylamine reaction or isocyanide test and is used as a test for primary amines.
ii) Aniline on diazotisation gives benzene diazonium chloride. This on warming with water gives phenol.
b) i) By Hoffman bromamide degradation reaction- when an amide is treated with bromine in an aqueous or ethanolic solution of sodium hy-droxide at about 343 K, an amine with one carbon less than that present in the amide is formed.
![]()
ii) By reduction – An amide on reduction with LiAIH4 or Na and ethanol an amine having the same number of carbon atoms as that in the amide is formed.
Question 1.
a) Amines are basic in nature. (March – 2013)
Arrange the following compounds in the increasing order of their basic strength.
NH3, C6H5NH2, CH3-NH2, (CH3)2 NH, (CH3)3N.
b) How will you convert aniline (C6H5NH2) to chlorobenzene?
Answer:
a) In aqueous solution when R = CH3 basic strength increases in the order
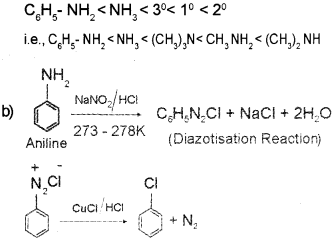
Question 1.
Amines can be considered as derivatives of ammonia. (Say – 2013)
a) Arrange the following in the increasing order of their basic strength.
C6H5NH2, C2H5-NH2(C2H5)2NH, NH3.
b) Represent a reaction of explain the basic character of aniline.
c) Name the reagents used in Hoffmann bromamide reaction.
d) What is the significance of the above reaction?
e) Give one chemical test to distinguish between methyl amine and diethyl amine.
Answer:
a) C6H5NH2 < NH3 < C2H5NH2, (C2H5)2NH
b) Aniline, being basic reacts with hydrochloric acid to form anilinium chloride salt.
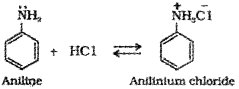
c) An amide, bromine, aqueous or ethanolic solution of NaQH.
d) It is a method for preparation of primary amines. The amine so formed contains one carbon less than that present in the amide.
e) Hinsberg’s test – When methyl amine (1° amine) is treated with Hinsberg’s reagent (benzene suiphonyl chloride), N-Methylbenzene suiphonamide is formed which is soluble in alkali due to the presence of acidic hydrogen.
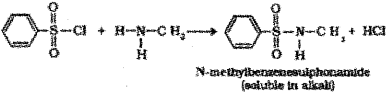
When dimethyl amine (2° amine) is treated with Hinsberg’s reagent N,N-dimethyl benzene shlphonamide is formed which is insoluble in alkali due to the absence of acidic hydrogen.
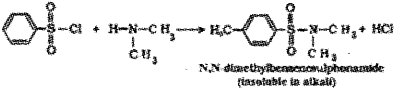
Question 1.
a) Write a method of preparation of primary amines. (March – 2014)
b) Describe a chemical reaction given only by primary amines.
c) What is diazotisation?
Answer:
a) Reduction of nitriles – Nitriles on reduction with LiAIH4 or catalytic hydrogenation produce primary amines.
OR
Reduction of amides with LiAIH4 produce primary amines.
OR
Pthalimide on treatment with ethanolic KOH forms potassium salt of phthalimide which on heating with alkyl halide followed by alkaline hydrolysis produces the corresponding primary amine.
b) Carbylamine reaction / Isocyanide test – Aliphatic and aromatic primary amines on heating with chloroform and ethanolic potassium hydroxide form isocyanides or carbylamines which are foul smelling substances. Secondary and tertiary amines do not show this reaction. This reaction is known as carbylamine reaction or isocyanide test and is used as a test for primary amines.
R-NH2 + CHC3 + 3KH R-NC + 3KCI + 3H3O
c) Conversion of primary aromatic amines into dia zonium salts by reaction with nitrous acid is called diazotisation.
Question 1.
a) Amines are basic. Arrange the following amines in the increasing order of basic strength (Say – 2014)
CH3 NH2, (CH3)2, NH, (CH3)3N, C6H5NH2.
b) Two well known reactions are given below: Suggest the main product of each reaction. Also give the name of each reaction.
Answer:
a) In gas phase:
(Hoffmann bromamide degradation reaction)
Question 1.
Amines are classified as primary, secondary and tertiary. (March – 2015)
a) Write the IUPAC name of the following compound: NH2 – (CH2) – NH2
b) Which is stronger base – CH3NH2 or C6H5NH2? Why?
Answer:
a) Hexane-1,6-diamine
b) CH3NH2 is a stronger base than C6H6NH2.
Alkyl amines are stronger than aniline. This is because the unshared electron pair on nitrogen atom to be in conjugation with the benzene and thus making it less available for protonation.
CH3NH2, due to electron releasing nature of the CH3– group, it pushes electrons towards nitrogen and thus makes the unshared electron pair more available for sharing.
Question 1.
a) Aromatic and aliphatic amines are basic in nature like ammonia. Arrange the following compounds in the increasing order of their basic strength: (Say – 2015)
CH3NH2,(CH3),NH,NH3,C6H5 -NH2
b) How will you carry out the following reactions?
i) Hoffmann bromamide reaction
ii) Carbylamine reaction (Chemical equations not required)
Answer:
a) C6H5-NH2<NH3<CH3NH2<(CH3)2NH
b) i) Hoffmann bromamide reaction – When an amide is treated with bromine in an aqueous or ethanolic solution of NaOH an amine with one carbon atom less than that present in the amide is formed. In this degradation reaction, migration of an alkyl or aryl group takes place from carbonyl carbon of the amide to the nitrogen atom.
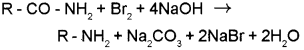
ii) Carbylamine reaction – Aliphatic and aromatic primary amines on heating with chloroform and ethanolic KOH form isocyanides or carbylamines which are foul smelling substances.
R – NH2 + CHCI3 + 3KOH → R – NC + 3KCI + 3H2O
Question 1.
Amines are classified as primary, secondary and tertiary amine. (March – 2016)
a) Represent the structure of secondary and tertiary amine.
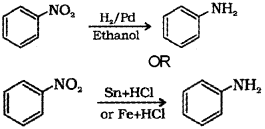
b) How will you convert nitrobenzene to aniline?
c) Aniline does not undergo Friedel-Crafts reaction. Why?
Answer:
b) Nitrobenzene on reduction by passing hydrogen gas in the presence of finely divided nickel, palladium or platinum and also by reduction with metals(Sn or Fe) in acidic medium gives aniline.
Or, the chemical equation:
c) Aniline does not undergo Friedel-Crafts reaction (alkylation and acetylation) due to salt formation with aluminium chloride, the Lewis acid, which is used as a catalyst. Due to this, nitrogen of aniline acquires positive charge and hence acts as a strong deactivating group forfurther reaction.
Question 1.
Amines are basic in nature. (Say – 2016)
a) Arrange the following compounds in the increasing order of their basic strength
NH3,C2H6NH2, C6H5NH2, (C2H5)2 NH
b) How will you convert aniline to chlorobenzene?
Answer:
a) C6H5 – NH2 < NH3 < C2H5NH2 < (C2H5)2NH
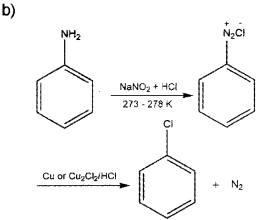
Question 1.
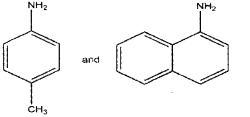
a) Classify the following amines as primary, secondary and tertiary (March – 2017)
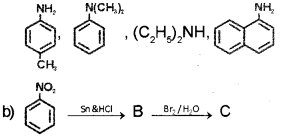
Identity the products B and C and write their formulae.
Answer:
a) Primary amines:
Secondary amine: (C2H5)2NH
Tertiary amine:
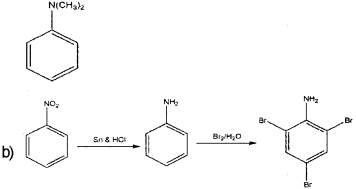
Product B is aniline and product C ¡s 2,4,6- tribromoaniline
Question 1.
a) The most basic compound among the following is (Say – 2017)
i) C2H5NH2
ii) C6H5NH2
iii) NH3
iv) (C2H5)2NH
b) Compound A is treated with Ethanolic NaCN to give the compound C2H5CN(B). Compound B on reduction gives compound C. Identify compounds A and C.
Answer:
a) iv) (C2H5)2NH
b) A-C2H5-X ethyl halide
C → C2H5-CH2NH2 propanamide
We hope the Kerala Plus Two Chemistry Chapter Wise Questions and Answers Chapter 13 Amines help you. If you have any query regarding Kerala Plus Two Chemistry Chapter Wise Questions and Answers Chapter 13 Amines, drop a comment below and we will get back to you at the earliest.
