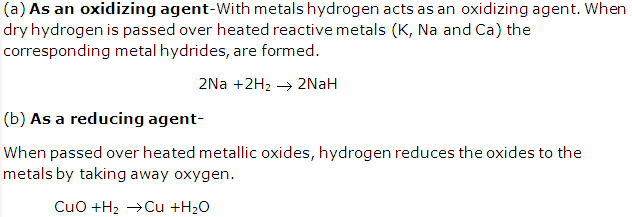Frank ICSE Solutions for Class 9 Chemistry – Study of the First Element – Hydrogen
PAGE NO :146
Solution 1:
- (a) Symbol of hydrogen is ‘H’ and its molecular formula is H2.
- (b) 2 H means 2 atoms of hydrogen while H2 means one molecule of hydrogen.
- (c) Lavoisier gave “Hydrogen” its name because it means water product.
Solution 2:
Hydrogen is found in free state as well as in combined state.
- In free state, it is found in traces in the earth’s crust and atmosphere. On the sun and bright stars, hydrogen is very abundant.It is suggested that the source of sun’s energy is fusion of hydrogen to helium.
- In combined state it is found in the compounds like water, acids, organic materials and minerals. Natural gas coming out from oil wells, coal mines and volcanoes invariably contains this gas.
Solution 3:
Hydrogen resembles the alkali in following manner-
- Electronic configuration – Hydrogen as well as alkali metals have one electron in their valence shell.
H (1) = 1 ; Li(3)= 2, 1 ; Na(11) =2, 8, 1 - Ion formation – Hydrogen loses one electron to form H+ ion like the alkali metals which form Li+, Na+, K+ etc.
- Valency electrons – Like alkali metals, hydrogen exhibit valency of one in its compounds , as it has only one electron in its outermost shell.
- Combination with non-metals – Like alkali metals, hydrogen combines with non-metals such as oxygen, chlorine and sulphur forming their oxides, chlorides and sulphides respectively.
- Reducing action – Like alkali metals, hydrogen is also a very good reducing agent.
Solution 4:
Hydrogen resembles the alkali in following manner-
- Electronic configuration – All the halogens have seven electrons in their outermost shell and need just one more electron to attain stable inert gas configuration. Similarly, hydrogen with one electron in its outermost shell requires one electron to attain a stable inert gas(Helium) configuration.
- Valency – Hydrogen and halogen both show a valency of one.
- Non-metals – Hydrogen is non-metallic like halogen.
- Atomicity – Hydrogen as well as halogens are diatomic gases, e.g.H2,Cl2.Br2 etc.
- Ion formation – Halogens have a strong tendency to gain an electron to form halide ions.In similar way, hydrogen shows tendency to gain one electron to form hydride ion (H–).
Solution 5:
Solution 6:

Solution 7:
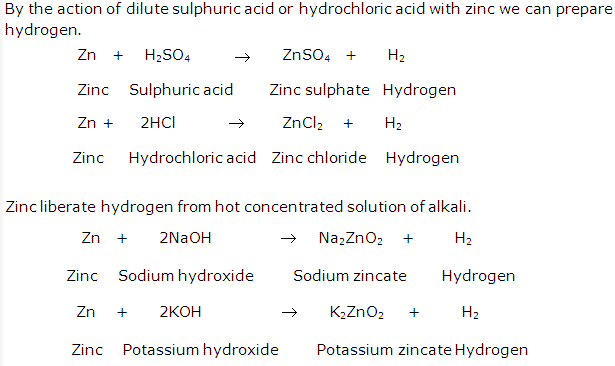
Solution 8:
Hydrogen gas obtained in the laboratory by dilute H2SO4 and granulated zinc have following impurities-
- Hydrogen sulphide (H2S),
- Sulphur dioxide (SO2),
- Oxides of nitrogen,
- Phosphine (PH3),
- Arsine (AsH3),
- Carbon dioxide, Nitrogen oxides,
- Water vapour
They are removed by passing through-- Lead nitrate solution-It absorbs H2S.
- Silver nitrate solution (AgNO3)-It absorbs PH3, AsH3
- Caustic potash (solid KOH)-It absorbs CO2, SO2, oxides of nitrogen
- Anhydrous calcium chloride or P2O5-It absorbs moisture.
Solution 9:
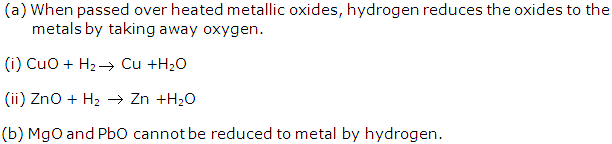
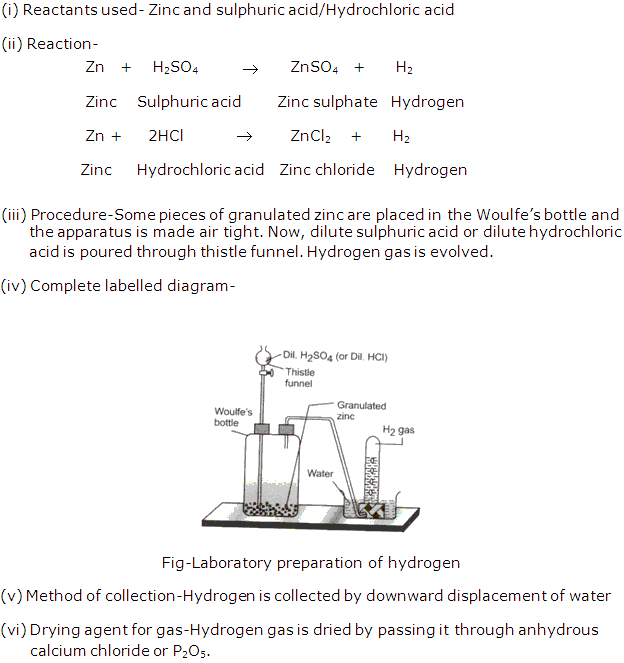
Solution 10:


PAGE NO :147
Solution 11:
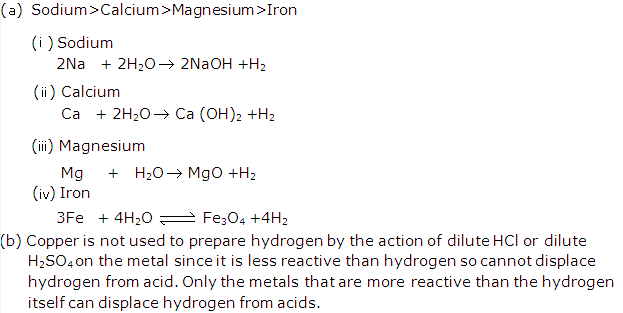
Solution 12:

Solution 13:
- (a) Nickel, hydrogen
- (b) above, dilute mineral
- (c) covalent, electronegative
- (d) CuO, hydrogen, water
- (e) CO, H2
- (f) alkali
- (g) nascent hydrogen
- (h) water
Solution 14:
- (a) False
- (b) False
- (c) True
- (d) True
- (e) True
- (f) True
- (g) True
- (h) False
Solution 15:


PAGE NO :148
Solution 16:
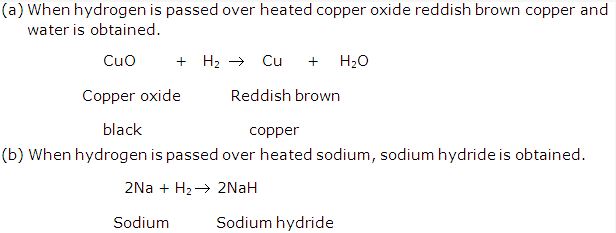
Solution 17:

Solution 18:

Solution 19:
Some pieces of zinc granules are placed in Woulfe’s bottle and the apparatus is made in air tight. Now dilute sulphuric acid is poured through the thistle funnel.
Observation: The reaction takes place at room temperature even without heating. A brisk effervescence with the evolution of gas is seen inside the bottle.
If burning candle is brought near the bubbles of hydrogen gas, they explode.This proves that bubbles were full of hydrogen gas and they move up in the air because they are lighter than air.
Solution 20:
- (a) Meteorological balloons – The low density and high lifting power of hydrogen made it useful in meteorological balloons used for studying air currents and weather conditions. However, due to its highly inflammable nature it has been replaced by helium which has a lifting only slightly less than that of hydrogen.
- (b) In metallurgy – Hydrogen acts as a very good reducing agent. It is used to obtain metals by reducing their oxides.
- (c) In fuel – Hydrogen has very high heat of combustions, therefore it is used as fuel in the form of coal gas, water gas and liquid hydrogen(for rocket propulsion).
- (d) In making fertilizers – A large quantity of hydrogen is used in the manufacture of ammonia by Haber process. Ammonia is used in manufacture of HCl and methyl alcohol.
Solution 21: