Analysing the electrolysis of molten compounds
- Electrolysis is a process whereby a compound is decomposed into its constituent elements when an electric current passes through an electrolyte.
- Michael Faraday was a pioneer in the field of electrolysis. He introduced the term electrolysis’ in 1834. The suffix ‘lysis’ is a Greek word, meaning ‘break down’.
- An electrolytic cell consists of a battery, an electrolyte that contains cations (positive ions) and anions (negative ions) and two electrodes.
- Classification of electrodes and their definitions are given in Table.
Term Definition Electrode A conductor in the form of a wire, rod or plate which carries electric current in and out of the electrolyte during electrolysis. Active electrode An electrode which takes part in chemical reactions during electrolysis.
Examples: metal electrodes such as copper, silver and mercury.Inert electrode An electrode which does not take part in chemical reactions during electrolysis.
Examples: carbon (graphite) and platinum electrodes.Anode An electrode which is connected to the positive terminal of the source of electricity in an electrolytic cell. Cathode An electrode which is connected to the negative terminal of the source of electricity in an electrolytic cell.
People also ask
- Why is an electrolyte able to conduct electricity while a Nonelectrolyte Cannot?
- Analysing the Electrolysis of Aqueous Solutions
- What does electrochemical series mean?
- How does a voltaic cell work?
- How is electrolysis used in the industry?
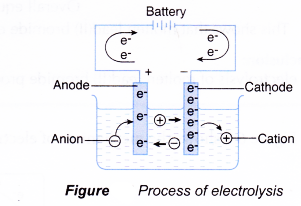
The process of electrolysis involves two stages.
(a) Stage 1: Movement of ions to the electrodes
- Cations (positive ions) move towards the cathode, the negatively-charged electrode.
- Anions (negative ions) move towards the anode, the positively-charged electrode.
(b) Stage 2: Discharge of ions
- Cations are discharged at the cathode by accepting electron(s) from the cathode, which has an excess of electrons.
- Anions are discharged at the anode by donating electron(s) to the anode, which has a lack of electrons.
- Electrons flow from the anode to the cathode through the connecting wire in the external circuit.
- When ions are discharged at the electrodes, they form atoms or molecules.
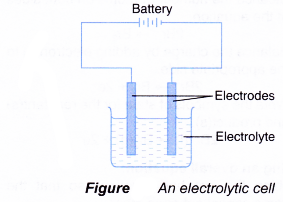
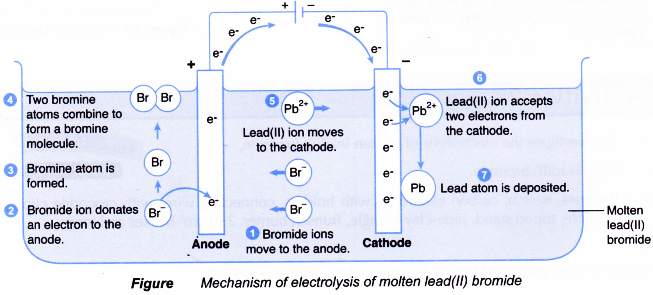
Figure shows the mechanism of electrolysis of molten lead(II) bromide.
Electrolysis of molten magnesium oxide:
- Molten magnesium oxide, MgO contains magnesium ions, Mg2+ and oxide ions, O2– that move freely.
- The Mg2+ ions move to the cathode while the O2- ions move to the anode.
- At the cathode: Each Mg2+ ion is discharged by accepting two electrons to form a magnesium atom, Mg.
Half-equation: Mg2+(l) + 2e– → Mg(s)
Thus, magnesium metal is formed at the cathode. - At the anode: The O2- ions are discharged by donating electrons to form neutral oxygen molecules, O2.
Half-equation: 2O2-(l) → O2(g) + 4e–
Thus, oxygen gas is released at the anode. - Overall equation:
2Mg2+(l) + 2O2-(l) → 2Mg(s) + O2(g)
Electrolysis of molten lead bromide experiment
Aim: To investigate the electrolysis of molten lead(II) bromide.
Material: Solid lead(II) bromide.
Apparatus: Batteries, switch, carbon electrodes with holders, connecting wires with crocodile clips, ammeter, crucible, tripod stand, pipe-clay triangle, Bunsen burner, 250 cm3 beaker and tongs.
Procedure:
- A crucible is filled with solid lead(II) bromide, PbBr2 until it is half full.
- The solid lead(II) bromide is heated until it is completely melted.
- The apparatus is set up as shown in Figure.
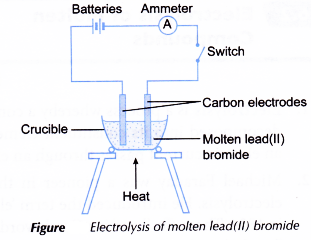
- The switch is turned on to allow electricity to pass through the molten lead(II) bromide for about 20 minutes.
- After that, the switch is turned off and both electrodes are taken out from the electrolyte. The molten lead(II) bromide is carefully poured into a beaker using a pair of tongs.
- All the observations are recorded.
Observations:
| Electrode | Observation |
| Anode | A brown gas with a pungent and choking smell is released. |
| Cathode | A shiny grey globule is found at the bottom of the crucible. |
The ammeter needle is deflected.
Discussion:
- The molten lead(II) bromide contains lead(II) ions, Pb2+ and bromide ions, Br that move freely.
- The Pb2+ ions move to the cathode while the Br– ions move to the anode.
- At the cathode: Each Pb2+ ion is discharged by accepting two electrons to form a lead atom, Pb.
Half-equation: Pb2+(l) + 2e– → Pb(s)
Thus, lead metal is formed at the cathode. - At the anode: The Br– ions are discharged by donating electrons to form neutral bromine molecules, Br2.
Half-equation: 2Br–(l) → Br2(g) + 2e–
Thus, bromine gas is released at the anode.
Overall equation: Pb2+(l) + 2Br–(l) → Pb(s) + Br2(g)
This shows that molten lead(II) bromide can be broken down to lead and bromine gas through electrolysis.

Conclusion:
The electrolysis of molten lead(II) bromide produces lead metal at the cathode and bromine gas at the anode.
